In a previous post, we looked at the colourless chemical compound known as isopropyl alcohol (IPA). With the formula C3H8O, it is a secondary alcohol that can be recognised by its strong odour.
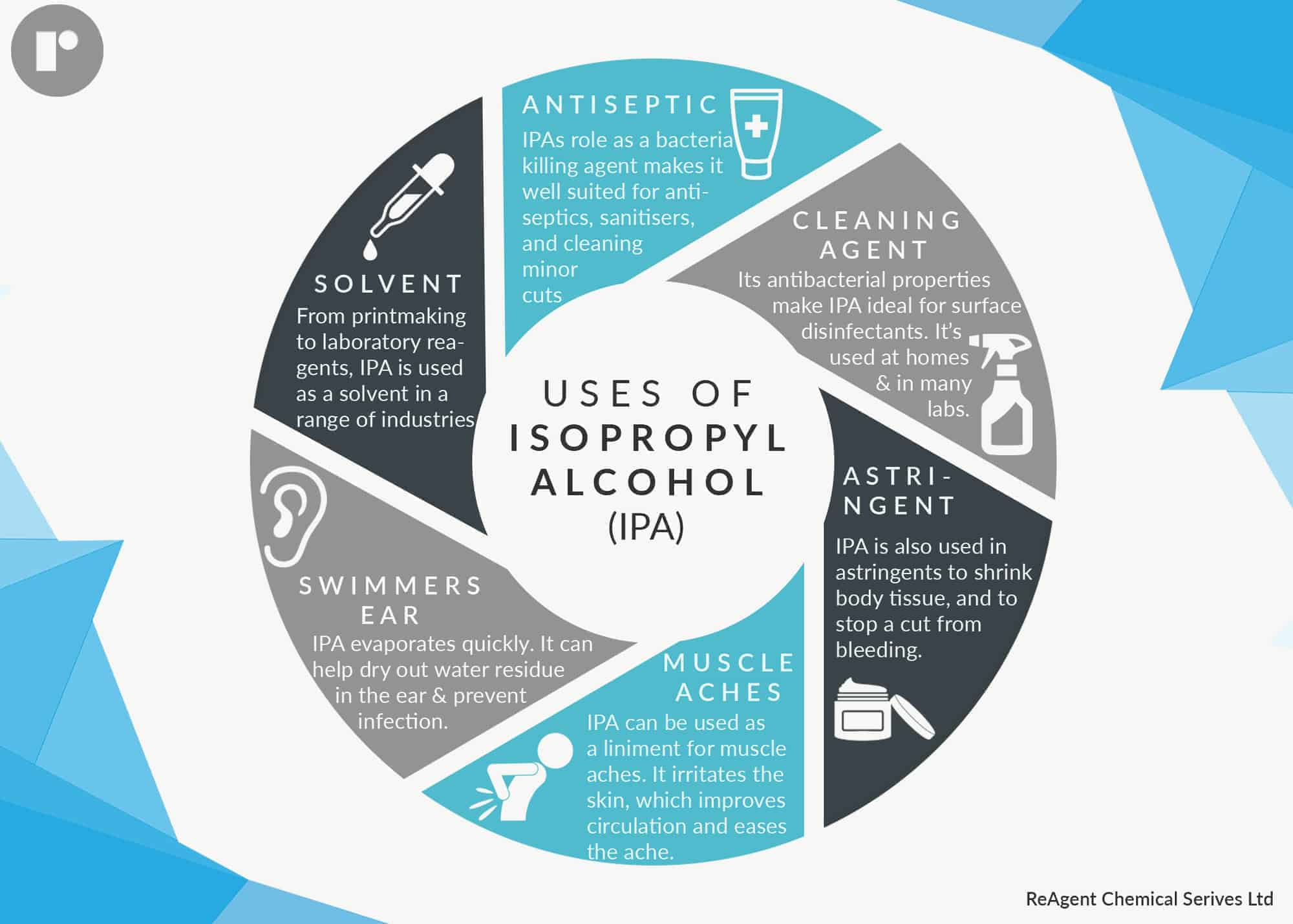
IPA is a volatile, flammable, and multipurpose product that has a range of uses in a variety of different industries. It’s most commonly used for its antibacterial properties, and how it is a staple in most home medicine cabinets. In this post, we explore the uses of isopropyl alcohol a little further to see how exactly it can be used.
In this post:
Antiseptic
Isopropyl alcohol, like many high proof ethanol products, is extremely antimicrobial against bacteria. This is why IPA is a popular antiseptic. It is what doctors swab your skin with before injections, what piercers use to prepare your skin for a piercing, and is found in many hand sanitisers. It is used in this way because it can kill and protect against surface bacteria. This is why it often used at home for minor cuts and scrapes. So why is this product effective as an antibacterial?
It comes down to IPA’s high alcohol concentration, which generally ranges between 70% and 99%. This classifies it as high proof, which means it is not safe to consume. When used as an antiseptic, a high concentration IPA solution – 70% is ideal – is able to penetrate the cell wall. It then permeates the entire cell and causes all proteins to coagulate. This kills the microorganism.
The presence of water in isopropyl alcohol solutions is vital for inhibiting the regrowth of pathogenic microorganisms because it helps to slow the speed of evaporation. This helps to increase surface contact time, which improves the antibacterial effectiveness.
Alcohol concentrations that are below 50% are not strong enough to be effective in this way – so don’t try using your 4.5% beer to clean a wound. Concentrations above 90% are similarly ineffective as they require longer contact time. This allows bacterial spores to lie in a dormant state without being killed. Therefore, an IPA solution of around 70% is your best bet in your fight against bacteria.
Cleaning Agent
Surface Disinfectant
Because of its antibacterial properties, as well as its capacity to act as a solvent, isopropyl alcohol is a great surface disinfectant. In fact, it is one of the active ingredients in many products that are used to disinfect hospital surfaces. A testament to IPA’s disinfectant abilities is the fact that it is on the Environmental Protection Agency’s “List of Antimicrobial Products Effective Against Mycobacterium Tuberculosis, Human HIV-1 and Hepatitis B Virus.”
If isopropyl alcohol is good enough to be used in hospitals and surgical hand scrubs, then it makes a great addition to the household cleaning cupboard.
Isopropyl alcohol is especially suited to cleaning glass surfaces, like windows or mirrors. This is because of its high volatility. By evaporating quickly, it leaves behind no streaks or residue, leaving you with a squeaky clean finish every time.
Household Solvent
IPA can be used in your home to clean and disinfect all surfaces, from countertop to toilet bowl. Its antiseptic properties means that it will kill lingering bacteria, and its ability to act as a solvent will enable it to dissolve sticky residues, grease, oils, etc. It can even erase permanent marker from surfaces by dissolving the mark to a liquid state, allowing you to easily wipe it away.
Electronic Components
From computer screens to contact pins in ROM cartridges, isopropyl alcohol is a favourite when it comes to cleaning electronic equipment. This is because it capable of thoroughly cleaning a component without leaving any streaks or any damage. It is commonly used in the electronic industry to clean magnetic tape, optical disc drive lasers, and IC packages. It can even be used on CDs and DVDs to wipe away fingerprints because it won’t damage the surface.
IPA can also come in aerosol form, which is optimal for electronic parts that have complex geometries or those that are difficult to access.

Astringent
Astringents are substances that can cause your body tissue, like pores, to tighten and contract. Astringents are regularly used on the face to make the skin look smoother by minimising pores. Isopropyl alcohol behaves as an astringent, and is a common ingredient in many skin toners and tightening formulas.
You can even do this at home by mixing equals parts of IPA and water and applying it to your face with cotton balls. However, because IPA is a skin irritant, you should always test your skin’s sensitivity to it first and ensure that you don’t leave it on for extended periods of time.
Astringents are also able to tighten capillaries that are losing blood following a minor cut. Therefore, IPA based astringents can also be used to stop minor bleeding.
Liniment for Muscle Aches
Because isopropyl alcohol is very drying, it is classified as a skin irritant. While you can use it as an antiseptic and astringent, this should only be done in small amounts to prevent skin damage. However, IPA’s propensity to irritate the skin does have one advantage.
A liniment is a liquid that is rubbed onto aching muscles in order to reduce pain and swelling. The fact that IPA behaves as a skin irritant means that it actually makes an effective liniment. This is because by irritating the skin, isopropyl alcohol is able to increase blood circulation to that area of the body. Improved circulation helps to ease pain and inflammation.
Swimmer’s Ear
For those of you who love swimming regularly, you may already be aware of, or have experience of the side effects of swimmer’s ear. For those of you who don’t, swimmer’s ear is an irritation and sometimes painful moment when water gets trapped deep in the ear canal.
Moisture that stays deep in the ear can be difficult to remove, can impede hearing, and even result in painful infections. This is because the residual moisture creates the perfect environment for infectious pathogens to proliferate.
Isopropyl alcohol is the swimmer’s best friend when it comes to preventing swimmer’s ear because it can protect against infection. Because IPA has a much higher vapour pressure at room temperature than water, it evaporates much quicker. Ultimately, drops of IPA in the ear will evaporate a lot faster than drops of water.
Therefore, by pouring a small of amount of IPA into the ear to mix with the water after swimming, the residual moisture will evaporate quickly. It will also dry out the ear and protect against infection.
Other Uses
We are only scratching the surface of the world’s most widely used solvent. Isopropyl alcohol is used in a wide range of applications that extend far beyond the above list. It is used in manufacturing acetone, glycerol, and isopropyl acetate; for de-icing your car’s windshield; as a laboratory reagent in a variety of chemical reactions, and even as a preservative and dehydrating agent in processes like sample preservation.
One of IPA’s most common application is as an industrial solvent, and can be used in a range of processes. Some its uses as a solvent include:
- Dilution and extraction in laboratory chemicals
- Paint stripping
- Flexography, lithography, and equipment cleaning in printmaking
- Cold-cleaning in electroplating
Drawbacks of Isopropyl Alcohol
There are many reasons why isopropyl alcohol is a favourite solution for homes and industries worldwide. However, as with every chemical product, it does have its drawbacks which should be kept in mind when you’re using it. Isopropyl alcohol is:
- An inhalation risk: constantly breathing in IPA’s fumes could result in dizziness, nausea and, in extreme cases, narcosis. It is important to use it in a ventilated area and to wear a mask if you’re using it for a prolonged period of time.
- A skin irritant: as we mentioned earlier, IPA can dry out the skin and cause redness. Always use small or diluted amounts if possible to avoid unnecessary skin exposure.
- Harmful to eyes: if you get IPA in your eyes, rinse with water immediately.
- A high proof alcohol: this means that it is not meant for human consumption. If ingested in large quantities, it could cause blindness and can even be fatal.
- Flammable: remember to keep IPA away from an open flame when in use. When used as a solvent, it is also important to bear in mind that high purity IPA has an open cup flashpoint of 11.7°C.
Whether you need isopropyl alcohol for laboratory, general, or analytical use, ReAgent has the batch that’s right for your business. Being accredited with the ISO 9001 quality standard means that you can rest assured that you will receive top grade IPA from a leading UK supplier. We also offer a variety of sizes, from 2.5L to 1000L, and a range of packaging materials so that your needs are met. Contact our expert team today to see what we can offer you.