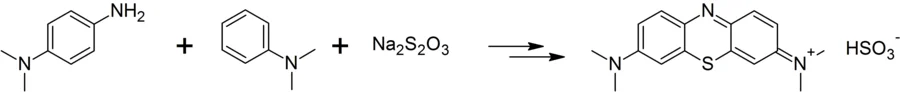Methylene blue has several medical applications, such as treating the blood disorder methemoglobinemia and counteracting the poisonous effects of potassium cyanide. In chemistry, it’s one of the various types of chemical indicators used in titration experiments and similar analytical methods. It’s used as a redox indicator, peroxide generator, water titrant, and sulphide indicator.
In this post:
What is the Chemical Name for Methylene Blue?
Methylene blue is known by many different names, most commonly methylthioninium chloride, but also Urelene blue, Provayblue, and Proveblue. Its IUPAC (International Union of Pure and Applied Chemistry) standardised name is 7-(dimethylamino)phenothiazin-3-ylidene]-dimethylazanium;chloride. Since this is descriptive, we can use it to deduce the chemical formula and chemical structure.
The name, specifically the chloride part, tells us that there is a presence of chlorine. However, the methylene group is structurally complex, and while we know it also contains sulphur and nitrogen, it would take an advanced knowledge of biochemistry to figure out the rest of the chemical composition solely from the IUPAC name.
Methylene blue is a derivative of phenothiazine, but is prepared by the oxidation of dimethyl-4-phenylenediamine in the presence of sodium thiosulfphte. The chemical equation for the reaction is shown below. As you can see, the sulfur is derived from sodium thiosulfate:
What is the Chemical Formula of Methylene Blue?
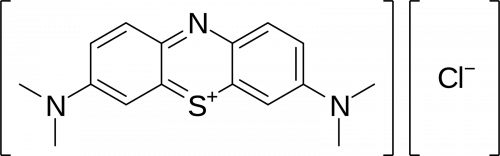
Methylene blue is a relatively complex organic compound that has three cyclic structures attached in a chain. Its chemical formula can be written simply as C16H18ClN3S. It has a molar mass of 319.85 g·mol−1 and becomes slightly acidic when dissolved in water.
But the molecular structure of the compound is actually a bit more nuanced, as shown in the illustration below. As you can see, sulphur and nitrogen are attached to the central ring, while chlorine is a separate group and is negatively charged:
What is the Function of Methylene Blue?
Methylene blue is a type of salt, meaning it has ionic bonds and a crystalline structure. As a salt, it dissociates as ions in water and turns the solution slightly acidic. While it mainly functions as a dye, it has other properties that make it suitable as a chemical indicator as well as a medication.
As a dye, methylene blue is very useful in the laboratory as an indicator of chemical change and as a biological stain for bacterial cells that are examined under the microscope. The dye highlights the contrasts, revealing details of the cells. The dye then attaches to protein receptors, allowing it to highlight the shape and structure of the cells.
These same chemical properties, which is having an affinity to stain cells, also make this salt useful as a medication and diagnostic tool in detecting certain abnormalities, like precancerous cells. While we know it has selective dye uptake effects on certain types of tissues, like nerve fibres, the exact mechanism isn’t yet fully understood.
What is Methylene Blue Used For?
Methylene blue is primarily used as a dye, but its dye properties also allow it to be useful in several other applications, such as in medicine, and in chemistry as a chemical indicator for various types of chemical reactions or properties.

Methemoglobinemia Treatment
Methemoglobinemia is a medical condition where methemoglobin is elevated in the blood. It’s a form of metalloprotein that’s similar to hemoglobin, but with an iron in the heme group that’s in the ferric (Fe3+) state, unlike the normal hemoglobin’s heme group, which is in the ferrous (Fe2+) state. The abnormal hemoglobin cannot bind with oxygen, which results in oxygen deficiency.
This condition is oftentimes caused by the ingestion of certain pharmaceuticals, toxins, or broad beans. Some of the symptoms include the following, all of which are due to insufficient oxygen levels in the blood:
- Headache
- Dizziness
- Difficulty breathing
- Nausea
- Muscle coordination problems
- Blue-coloured skin or cyanosis
- Seizures
- Heartbeat irregularities
Methylene blue and oxygen therapy is used to treat methemoglobinemia. The chemical pathway is through NADH or NADPH dependent methemoglobin reductase enzymes. The abnormal hemoglobin is reduced back to normal hemoglobin. However, high doses of this salt could also induce methemoglobinemia, meaning proper dosage is critical.
Redox Indicator
Redox, or oxidation-reduction reactions, involve the transfer of electrons from one species to another. When a chemical species is oxidised, it loses electrons but gains or combines with oxygen. Conversely, reduction is the gaining of electrons and the losing of oxygen. Redox reactions always go side-by-side. An example of this is the oxidation-reduction reaction between ferric oxide and carbon monoxide, as shown in the balanced chemical equation below: Methylene blue is a good redox reaction indicator: it’s blue when mixed with an oxidising agent, but turns colourless when mixed with a reducing agent.
Methylene blue is a good redox reaction indicator: it’s blue when mixed with an oxidising agent, but turns colourless when mixed with a reducing agent.
One of the traditional ways of demonstrating chemical kinetics in general chemistry is the blue bottle experiment. In this classical experiment, a solution is made from glucose, methylene blue, and sodium hydroxide. The solution container, typically a flask, is shaken, resulting in the oxidation of methylene blue. This turns the solution blue. The glucose then gradually reduces the chemical indicator, turning it colourless. The solution will again turn blue as the glucose is consumed in the reaction.
Watch a demonstration of this experiment:
Cyanide Antidote
The reduction capacity of methylene blue is very similar to that of oxygen, in that it can be reduced by components of the electron transport chain. Therefore, it can be used as an antidote for potassium cyanide poisoning. The method for this was first proven effective in 1933 by Dr. Matilda Moldenhauer Brooks.
Water Testing
Methylene blue can also be used to detect surfactants in water samples. For example, you could make a solution of methylene blue and chloroform in order to determine the presence of anionic surfactants, such as traces of detergents or emulsifiers.
What Are the Chemical Properties of Methylene Blue?
The key chemical properties of methylene blue are its colour changing ability and the fact that it becomes acidic when dissolved in water.
In a solution, methylene blue is slightly acidic and dissociates into its ionic constituents. An aqueous solution of this compound has a pH level of 6 at a concentration of 10g/l and at an ideal temperature of 25 °C.
Meanwhile, the colour change in methylene blue is because it loses electrons in the presence of oxidising agents. This makes it good at detecting oxygen in a solution.