One of the most important applications of chemistry is storing electrical energy for prolonged periods of time. Modern technological products like laptops, mobile phones, solar power, and hybrid cars would not have been possible without the invention of batteries, which are basically electrochemical storage devices that have two electrodes and an electrolyte.
In order for batteries to work, regardless of the type, there must be electrical potential between two materials (the electrodes) that are immersed in an electrolyte.
In this post:
Early History of Batteries
As early as 1749, Benjamin Franklin experimented with electricity using Leyden jars as linked glass capacitors. He was the first to use the term ‘battery’ to describe two or more similar objects functioning together. This term was then applied to voltaic piles, the first electrical batteries.
While Luigi Galvani discovered bioelectromagnetics in 1780 by touching legs of frogs using a different metal from the metal supporting the suspended legs, it wasn’t until 1800 that Alessandro Volta developed the voltaic pile, which consisted of copper and zinc disc pairs on top of each other. These were separated by a layer of cloth soaked in saltwater, acting as the electrolyte. Thus, the first true battery was born.
However, some may dispute the claim that Alessandro Volta invented the first battery, and would instead point to the so-called Baghdad battery as proof. However, the real purpose of this archeological artefact remains inconclusive and controversial.
What Are Electrochemical Cells?
Batteries can either be composed of just one electrochemical cell or several electrochemical cells that are connected in parallel or in series. The main difference between ‘in parallel’ and ‘in series’ is that a parallel connection increases the current or amperage, whereas a series connection increases the voltage.
An electrochemical cell is a unit that can be considered as a battery or storage of electricity in itself, but can also be a part of a single battery system, such as a car battery.
As the name implies, an electrochemical cell is a unit that produces electricity through chemical reactions. It involves electrodes (anodes and cathodes) and an electrolyte.
Electrodes are different conductive substances that have electrical potential between them. For example, in a dry cell battery, like those used in torches and TV remotes, the anode is the zinc inner case of the battery. Meanwhile, the cathode is a graphite rod.
Inside the battery, an electrolyte paste surrounds the graphite rod. This is so tightly packed that it touches the inner wall of the zinc casing. The paste is composed of carbon, ammonium chloride (NH4Cl), and manganese oxide (MnO2). Here’s an illustration:

In a battery, the anode refers to the negative electrode of a primary cell that releases electrons into the external circuit. This happens through the oxidation process. However, if the battery is rechargeable, the anode becomes the positive electrode during recharging.
Conversely, the cathode in a battery refers to the positive electrode receiving the electrons that flow from the anode. This is where the reduction reaction occurs when the battery is being used. If the battery is rechargeable, this electrode becomes the anode when the battery is being recharged.
What is the Conventional Cell Representation?
Below is another illustration of an electrochemical cell that’s typically seen in a laboratory setup. Again, it involves two electrodes and an electrolyte. The transfer of electrons from the anode to the cathode creates a chemical reaction that changes the electrolyte as the electrochemical cell is being discharged with a load, such as in a small light bulb:
As you can see in the illustration, as the anode (zinc) loses electrons, the cathode (copper) gains electrons. The electron flow can be written as half-cell reactions, which are more simplified so that you don’t need to draw the electrochemical setup.
How to Measure Half-Cell Potential
Over the course of almost three centuries, after Benjamin Franklin first conducted experiments on electricity using Leyden jar capacitors, many types of batteries have been invented. Some examples are nickel iron batteries, zinc-carbon dry cell batteries, lead-acid batteries, alkaline batteries, and lithium ion batteries.
Measuring the half-cell or electrode potential of batteries is crucial to battery design and efficiency. The standard hydrogen electrode is used as the standard measurement for half-cell potentials. The standard hydrogen electrode has 0.00V half-cell potential under the following standard conditions:
- Solution with a concentration of 1.0 moldm3
- Temperature of 289K
- Pressure of 100kPa
The half-cell consists of hydrochloric acid, hydrogen gas, and platinum electrodes.
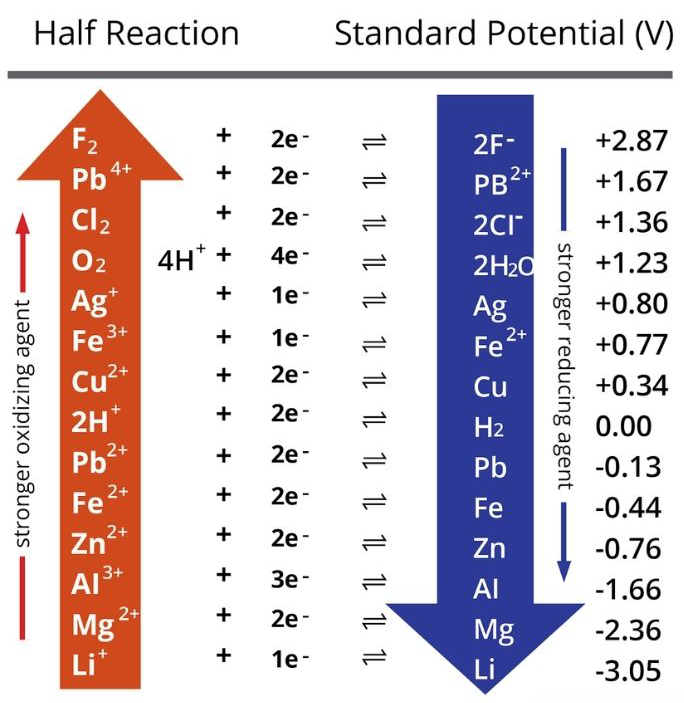
The table below shows the standard potentials of cathode half reaction at standard conditions. Electrodes that are very positive are better oxidising agents, and oxidise those that are less positive. For more comprehensive comparisons, click here.
What Are the Different Classifications of Commercial Cells?
Although there are many types of batteries, there are only three main categories of electrochemical cells that are used as base units of commercial batteries. Commercial cells are classified as rechargeable, non-rechargeable, and fuel cells:
- Rechargeable cells: The chemical reactions that occur in these cells are reversible. The electrode potential can be restored after the cell is discharged. Some examples of these include lead-acid cells and lithium ion cells. For example, mobile phones, laptops, and tablets are reliant on lithium batteries as a power source.
- Non-rechargeable cells: The chemical/ionic reactions that occur in non-rechargeable cells are irreversible. The most common example of this is the carbon-zinc dry cell, commonly used in flashlights, TV remotes, and portable radios.
- Fuel cells: A fuel cell can continually supply electricity without needing to be recharged, as long as there is a steady supply of fuel. The most common example of this is the hydrogen fuel cell. Hydrogen is oxidised in the anode terminal, which produces water and two extra electrons. Meanwhile, the oxygen in the cathode terminal receives two extra electrons and undergoes a reduction reaction. These reactions result in electrode potential that allows the flow of electrons, or electrical current.