As you might expect, purified water is exactly what it says – water that has been purified to varying degrees. It can be made via many different processes, and is used in a range of industries.
From purified bottled water to water that is pure enough for laboratory applications, this article will discuss the science behind purified water.
In this post:
Purified Water: A Brief Overview
Purified water isn’t necessarily 100% pure. Regular tap water, for example, must undergo a process of purification in order for it to become drinkable.
This is not just done naturally, like when water passes through springs or underground aquifers, but involves a series of mechanical filtrations and chemical treatments. These purify the water from harmful amounts of mineral residues, heavy metals, and soil particles. Purification also removes pathogens, such as bacteria, viruses, fungi, amoeba, microscopic worms, and even parasitic eggs from water.
Municipal water undergoes several steps of purification before it becomes potable and is distributed to the public. Here are the four steps:
- Step 1 – Coagulation and flocculation: At this stage, negatively charged particles in water are filtered out by adding in positively charged chemicals. The particles then coagulate into larger particles called floc.
- Step 2 – Sedimentation: The larger and heavier particles that clumped into flocs settle to the bottom of the water supply treatment facility. They’re then easily separated from the water.
- Step 3 – Filtration: The water then passes through several filtration systems that consist of sand, charcoal, and gravel. This process removes microscopic contaminants, such as bacteria and chemicals.
- Step 4 – Disinfection: Chemical disinfectants like chlorine are added to the purified water in order to kill any pathogens in it.

Different Grades Of Water
Water can be purified to either completely, or partially, remove organic and inorganic impurities. This makes it suitable for a wide variety of uses, depending on which purification method is used. To gauge the level of purity, water is classified into grades.
For laboratory applications, the International Organisation for Standardisation (ISO) has specified three grades of water: Grade 1, which is the purest, Grade 2, and Grade 3, which is the least pure. Each of these grades of water has its own specifications, according to ISO 3696:
- Grade 1: Otherwise known as ultrapure water, this is the purest grade of water. As such, it’s used in very sensitive laboratory procedures, such as culturing mammalian cells.
- Grade 2: Commonly known as purified water, this is used in general laboratory applications, such as preparing buffers, where the purity level is important, but not crucial.
- Grade 3: Also called primary grade water, this is used in procedures that are considered to be non-critical, like rinsing laboratory glass equipment or feeding autoclaves.
What Methods Are Used To Purify Water?
There are many methods that are used to purify water. The most common method is simply boiling it, which kills bacteria and viruses. While this method is effective for purifying ordinary drinking water, it’s nowhere near effective enough to be used in laboratory or medical applications.
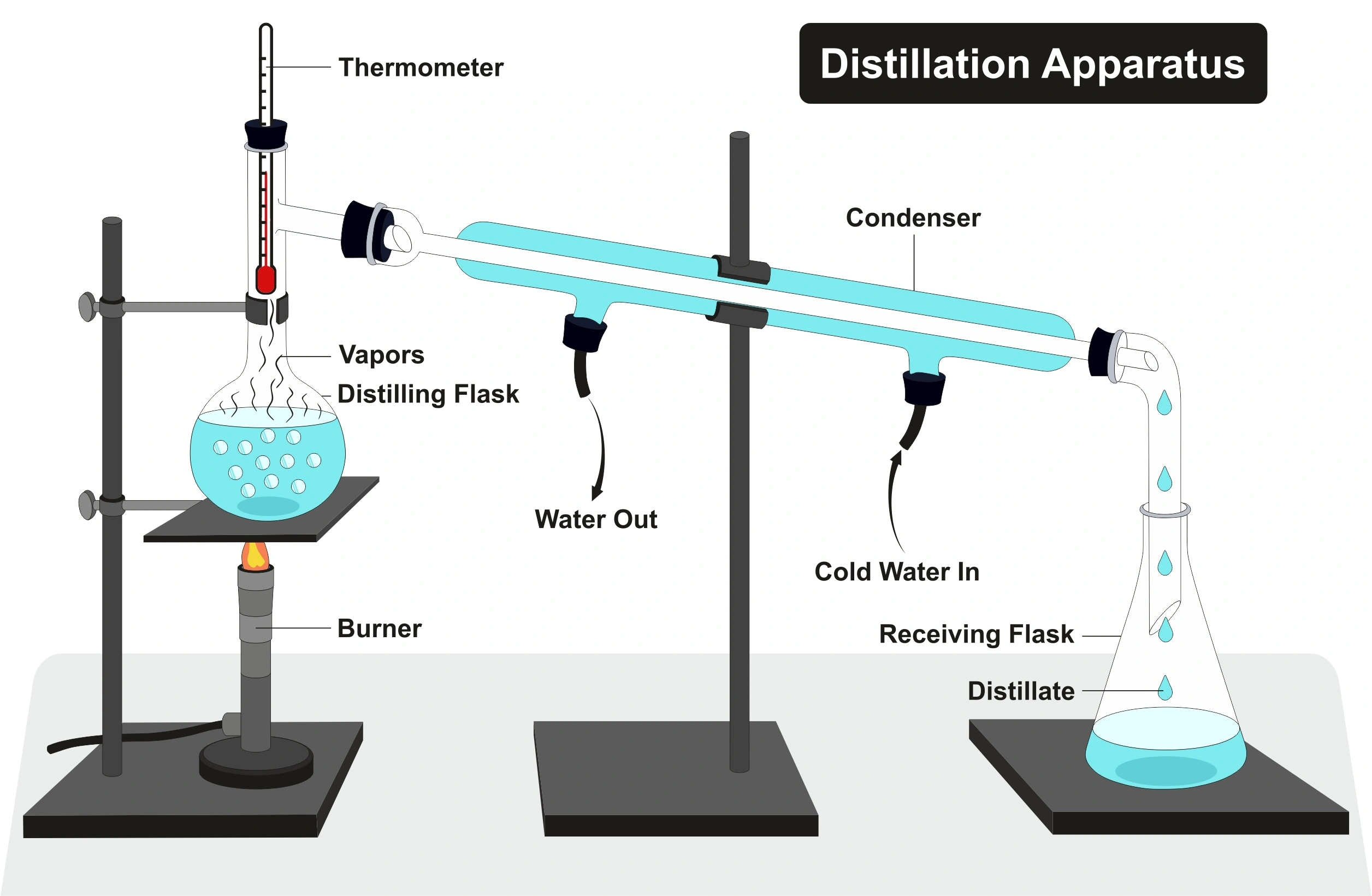
When higher purity water is needed, distillation tends to be the go-to method of purification.
Distillation has been used since ancient times to purify the alcohol content in fermented alcoholic liquids, transforming it into spirits. Nowadays, distilled water is the most common form of purified water on an industrial scale. The process basically involves boiling water in one container and allowing it to precipitate and condense in another.
Aside from distillation, the other methods of purifying water include:
- Deionisation
- Reverse osmosis
- Carbon filtering
- Microfiltration
- Ultrafiltration
- Ultraviolet oxidation
- Electrode ionisation
Commercially available purified water is processed using combinations of two or more of these purification methods.
As a result, it’s now possible to have water that has barely any contaminants in it – they can be so low, in fact, that they can’t be measured in parts per billion, or even parts per trillion.
Reagent grade or laboratory grade purified water has several applications. In terms of large-scale applications, highly purified water is mainly used in the production of medicines. Meanwhile, the industrial production of lower-grade purified water is largely used in the beverage industry.
What Is The Process Of Purifying Water Called?
The process of purifying water goes by many names, some which we’ve just mentioned, like distillation, deionisation, and ultrafiltration. What the purification process is called depends on what method is being used.
Although there are various processes and steps involved in purifying water, the main mechanisms function in the same way: they remove impurities from the water. The various steps involved in water purification can be classified into four categories:
- Physical processes: These include filtration in various stages, sedimentation, and water distillation. One, or a combination of various processes, are used here.
- Biological processes: These involve the removal of organic and biological contaminants in water. Sand filters and biologically active carbon are also used in the process.
- Chemical processes: Some examples of chemical processes are chlorination and flocculation, wherein chemicals are applied to remove or kill contaminants.
- Radiation exposure processes: During these types of processes, water is irradiated to kill pathogens like bacteria and viruses. The most common method used is ultraviolet light exposure.
What’s The Difference Between Purified And Filtered Water?
Generally speaking, filtered water is one type of purified water. However, its purity depends on the level of filtration that has been used and how many contaminants have been filtered out. Remember, it doesn’t count as purified water if the level of contaminants are not within the standard range.
The main difference is that filtration is typically used to remove solid particles, while purification is used to eliminate all forms of contaminants.
For example, muddy water can be filtered in various ways, such as through reverse osmosis and sand-carbon filtration. But if some sediment or other particles still remain, it’s not yet purified. Water has to undergo various stages of filtration and purification before it’s considered pure.
What Is The pH Of Purified Drinking Water?
The pH level of purified water depends on its type and the processes it’s undergone. Here’s a list of the various types of purified water and their corresponding pH levels:
- Tap water has an average pH of 7.5
- Common bottled water has a pH of between 6.5 to 7.5
- Bottled water marketed as alkaline water has a pH between 8 and 9
- Water from distilled reverse osmosis has a pH of between 5 and 7
It’s a misconception that water that has been properly purified, either through distillation or deionisation, has a neutral pH of exactly 7.
While these purification methods do remove most pH swayers, like ions, minerals and other contaminants, the water they yield will have a slightly acidic pH. This is because it absorbs carbon dioxide from the atmosphere, which brings its pH down to a comfy 5.8 or 5.5.

Social Impact Of Water Purification
Water purity is a serious issue that is directly related to health and environmental sustainability. Contaminated water is very dangerous because it carries pathogens, such as the bacterium species that causes cholera.
Other diseases contaminated water results in include:
- Dysentery
- Typhoid fever
- Schistosomiasis
- Polio (infantile paralysis)
Globally, it’s estimated that around 2.2 billion people lack access to clean drinking water. Many natural sources of freshwater, such as rivers, ponds, and lakes, are unfortunately not suitable for drinking due to pollution. Even water that comes from springs and aquifers can be contaminated by pathogens and toxic substances that need to be filtered out.
Many communities simply do not have the resources to process contaminated water into drinking water, and commercially available bottled water products are sometimes too expensive for many people living in impoverished counties.
However, the process of purifying water can be made more affordable for communities and households that need it the most. For instance, the water filter straw could potentially have a significant impact on the lives of millions of people in third world countries.











