Muriatic acid is one of the most common acids used in industrial and household applications.
This article provides more information about this acid, including its chemical properties, its various uses, and why it has come to be known by so many names.
In this post:
What Is Muriatic Acid Also Known As?
Muriatic acid is more famously known as hydrochloric acid, and it normally appears as a colourless aqueous solution. However, some bottled versions of this products may have a yellow colouring. This is actually an indication of the presence of impurities, such as chlorine gas, iron, and organic substances.
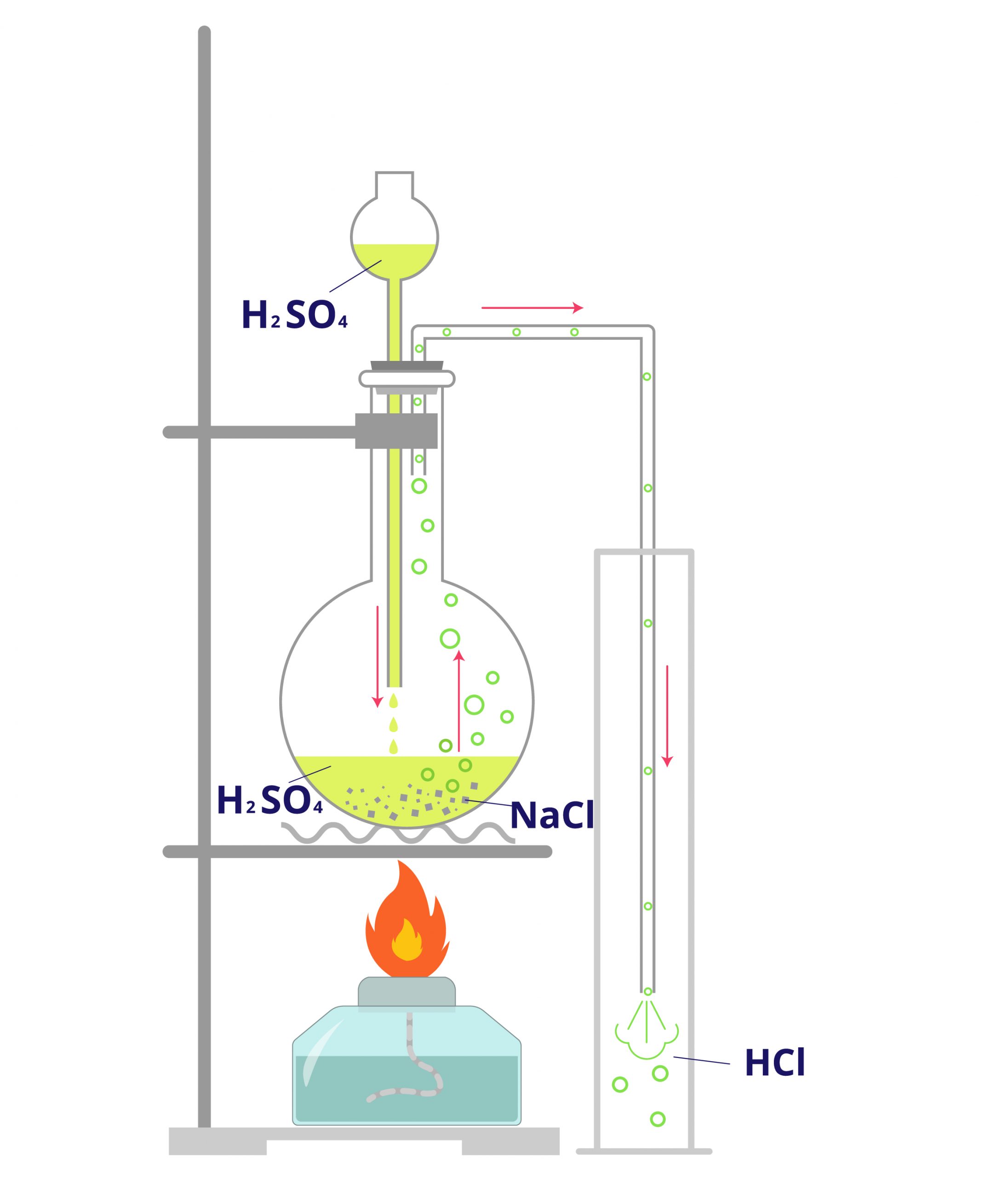
Hydrochloric acid is produced when hydrogen chloride is dissolved in water. Historically, it was produced by mixing solutions of green vitriol or iron(II) sulphate with rock salt or halite – which is basically sodium chloride. In the modern production of muriatic acid, sodium chloride and sulphuric acid are mixed to produce hydrochloric acid and sodium sulphate.
Its properties and uses meant that hydrochloric acid became known by many aliases. For example, the word ‘muriatic’ is derived from ‘brine or salt’ because of its use as a pickling agent. This led to it being known as spirits of salt or acidum salis by European and Latin alchemists, referencing its behaviour as a salt acid.
Another name for muriatic acid is hydronium chloride, which simply references the aqueous solution of the salt form of the acid, a.k.a. hydrogen chloride. This is because in an aqueous solution, hydrogen and chlorine become ions.
Muriatic acid is chemically identical to hydrochloric acid. The main difference is the level of purity. The level of purity includes the proportion of the acid as solute with the solvent, which is water, as well as other chemical residues in the solution, like metal ions. With that in mind, commercially available muriatic acid usually has a pH level of between one and two.
You can decrease its acidity by diluting it with water, and you can do this according to your intended purpose, whether it’s for toilet cleaning or neutralising swimming pool water. Commercially available hydrochloric acid has a lower grade classification (between 14.5% and 28% hydrochloric acid) compared to reagent grade hydrochloric acid, which has a proportion of 37% hydrochloric acid and 63% water.
Muriatic acid is as corrosive as the gastric juice in the stomach, with a pH range of between 1.5 and 3.5. However, stomach acid is actually only 0.5% hydrochloric acid. Its other major non-organic components are electrolytes like potassium chloride, phosphate, bicarbonate, and sodium chloride. Our stomach acid also contains organic substances, like mucus and pepsins.
The pH level of an acid is not the standardised measure of its strength, since this factor may vary depending on the proportion of the solute and solvent. Hydrochloric acid is considered as one of the seven strong acids, not because of its pH level but because of its dissociation constant. The dissociation constant refers to the readiness of ions to separate in aqueous solutions.
The other six strong acids that have this are:
- Nitric acid
- Sulfuric acid
- Hydrobromic acid
- Hydroiodic acid
- Perchloric acid
- Chloric acid
As a rule of thumb, a strong acid completely dissociates into constituent ions in a one molar solution of that acid. Weaker acids, like acetic acid, only partially dissociate in a solution.

What Are The Uses Of Muriatic Acid?
Muriatic acid is a highly corrosive substance even as a diluted form. It can dissolve iron, and it violently reacts with other metals, like aluminium and magnesium, as well as bases, like sodium hydroxide.
These reactions are highly exothermic, and release large amounts of heat energy during the process. The high reactivity of muriatic acid makes it very useful in a wide range of industrial and household uses.
This acid is a common household cleaner, and is typically used for cleaning toilets and tiled floors. It’s also used in maintaining the pH balance of swimming pools. As well as all this, hydrochloric acid can be used either as a precursor or as an ingredient in manufacturing other chemicals, like disinfectants and slimicide (an anti-slime agent).
This acid is essential in the large-scale production of vinyl chloride, which is used in manufacturing polyvinyl chloride (PVC) plastic. Muriatic acid is also an essential precursor for producing polyurethane foam.
Here are the top industrial applications of muriatic acid:
- Steel production: Pickling steel is one of the most important industrial applications of hydrochloric acid. It’s basically about removing rust or iron oxide from steel before it’s subjected to other processes, like extrusion. A concentration of 18% muriatic acid is used for pickling.
- Organic compound production: Hydrochloric acid is also essential in the large-scale production of organic chemicals like vinyl chloride and dichloroethane, which are used in manufacturing PVC plastics. Here, the acid is used for producing bisphenol A for polycarbonate and ascorbic acid. Hydrochloric acid is also essential in producing various pharmaceutical products, and behaves either as a reagent or precursor for organic chemicals.
- Manufacture of inorganic compounds: By allowing muriatic acid to react with various base substances, a range of new compounds can be formed. Two important products of such reactions are iron(III) chloride and polyaluminium chloride. These compounds are used in water treatment, particularly as flocculation and coagulation agents, allowing pollutants in sewage water to settle and be removed. Another important inorganic compound from the reaction of hydrochloric acid with base substances is zinc chloride. This has an important role in producing batteries and galvanising metals.
How Is Muriatic Acid Made?
The industrial demand for muriatic acid, or hydrochloric acid, is growing. On an industrial scale, hydrochloric acid is manufactured by dissolving hydrogen chloride in water. Several precursors and ways of producing hydrogen chloride exist. Its large-scale production is tied with other industrial productions, like that of hydroxide, hydrogen, and chlorine.
Muriatic acid can be made by the direct reaction between hydrogen and chlorine. However, the most common process of producing hydrochloric acid is through the chemical reaction between sulphuric acid and sodium chloride. The balanced chemical equation below shows the reaction:
NaCl + H2SO4 → HCl + NaHSO4
How To Handle Muriatic Acid
Muriatic acid, even at low concentrations, is corrosive. It can potentially burn your skin or damage your eyes. The fumes can also be toxic when they react with other substances.
With that in mind, it’s crucial to take some precaution when handling muriatic acid, either as a chemical reagent, a precursor in a laboratory setting, or even just simply as a cleaning agent. In all cases, it’s important to check the guidelines and wear the following PPI (personal protective equipment):
- Goggles
- Rubber gloves
- Apron or lab gown
- Breather mask or vapour respirator
Although hydrochloric acid is part of the gastric acid in the stomach, it’s still toxic when ingested because of its impurities and corrosivity – it can even burn your oesophagus and lungs. So, you should always take precautions when handling muriatic acid.